AlnnoVent AVB-100
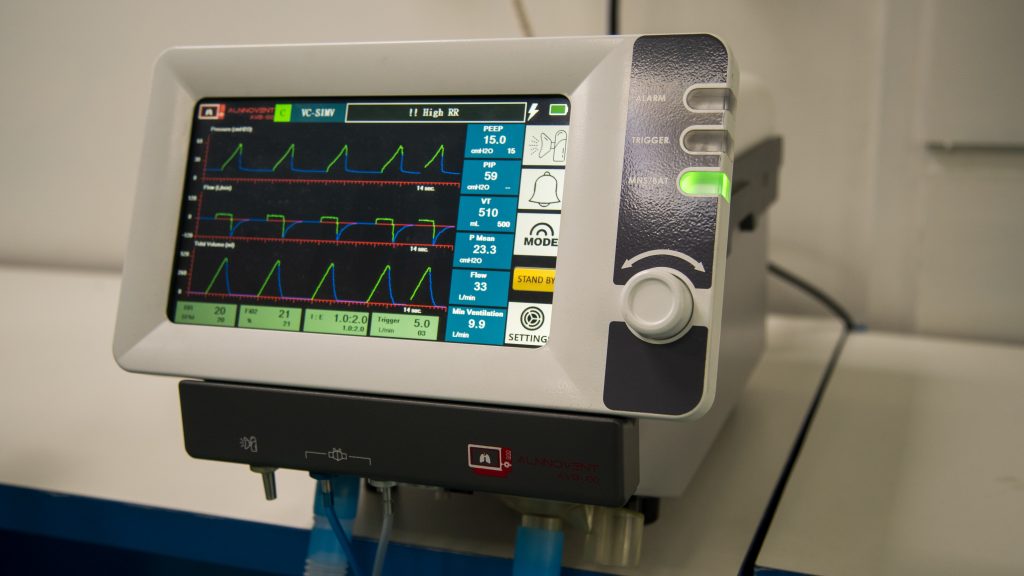
The AlnnoVent is designed as a ventilator for adult patients weighing between 30 kg and 300 kg. This ventilator is intended for use by trained technicians under the supervision of physicians.
Mechanical ventilation procedure
is performed in patients with
respiratory failure with symptoms
broadly categorized as the
inability to meet the body’s need
for oxygen delivery or carbon
dioxide removal. Ventilator functions to deliver air usually with an elevated oxygen content via an
endotracheal tube to the patient’s lungs, facilitating the exchange of oxygen and carbon dioxide.
Airway protection, treatment of hypoxemic respiratory failure (low blood oxygen), treatment of
hypercapnic respiratory failure (elevated carbon dioxide in the blood), or treatment of a combined
hypoxic and hypercapnic respiratory failure are indications for mechanical ventilation.
The AlnnoVent has an edge over other ventilators developed in Pakistan, our device is in the
process of clinical trials. Whereas other devices such as i-Live by PAEC have passed DRAP
approval, they are not moving towards manufacturing and commercialization of their product.
The Alsons Group was also repeatedly mentioned in the news in December 2020 – April 2021.
As stated in Dawn news article; AlnnoVent developed by Alsons Group Karachi was cleared by
PEC and its application for National Bioethics Committee (NBC) was under process for a clinical
validation study.
As mentioned in the News Update’s article, The Ministry of Science and Expertise held the
nationwide launch of the Alnno-Ventura HFNO system, a UK certified, a subtle respiratory system
developed in Pakistan by The Alsons Group. The Alnno-Ventura follows the designs and specs
of the UCL Ventura manufactured below license and directions from the UCL Institute of
Healthcare Engineering within the UK.
OBJECTIVE
Increase awareness of locally manufactured ventilators in Pakistan initially and later
target other LMIC’s.
• Build connections with healthcare professionals and create better distribution
networks
• Increase the overall presence of AlnnoVent on social media.
